Head of group
Prof. Květoslav Růžička, Ph.D.
b kvetoslav.ruzicka![]() vscht.cz
vscht.cz
e +420 22044 4116
d A131
Team members
Prof. Michal Fulem, PhD.
Vojtěch Štejfa, PhD.
Alex Mathers
Václav Pokorný
Research topic
Description of the vapor-liquid and vapor-solid equilibria for i) high-boiling substances classified as potential contaminants; ii) organometallic compounds used as MOVPE precursors; iii) biogenic compounds (terpenes, fragrances, H-bonding compounds, infochemicals). This includes
a) experimental determination of vapor pressure in low-pressure region (below 1 kPa);
b) calorimetric determination of heat capacities and enthalpies of phase transitions for solids and moderately volatile liquids (temperature range from 2 K to 600 K);
c)state of the art ab-initio calculations of ideal gas thermodynamic properties;
d) simultaneous correlation (SimCor) of vapor pressures and thermal derived quantities, which allows evaluation of vapor pressure (and vaporization/sublimation enthalpies) in the region of extremely low vapor pressures, where direct determination is not feasible;
e) Development, validation and application of ab initio computational methodologies for calculations of thermodynamic properties, phase diagrams and polymorphism of molecular crystals;
f) Molecular-dynamics simulations and calculations of thermodynamic and structural properties of ionic liquids.
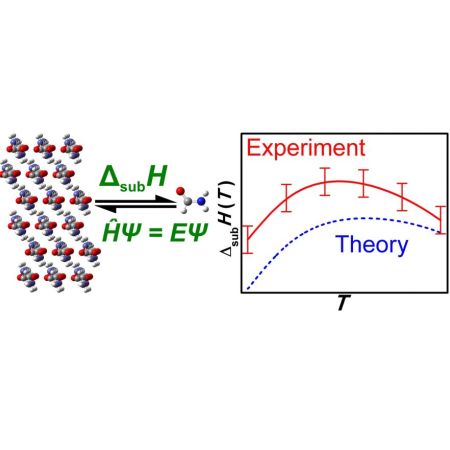
Sublimation enthalpies obtained by SimCor are used as a benchmark for development of ab initio methodologies leading to reliable lattice energies and prediction of sublimation equilibria.
Calorimetry is used also for studying novel inorganic materials used for fabrication of semiconductors; polyaromatic hydrocarbons and bitumens; compounds of pharmaceutical interest
